|
The first level is filled and can not accommodateany more electrons. When the third electron is to be placed it must go into thesecond level. In multi-elecron atoms the degeneracy of theenergy of the sublevels is lost. In the hydrogen atom the sublevels in each principle levelare degenerate. Notice that there hasbeen a change in the relative energies of the 2s and 2p orbitals.This is an important point that must be addressed at this point. Theenergy diagram for helium is shown as here. So while hydrogen has the electron configuration of 1s 1,helium has the electron configuration of 1s 2. It turns out that the energy required to accommodatetwo electrons in the 1s orbital is significantly less than theenergy required to place the second electron into the higherenergy n = 2 level. So the secondelectron could go into the 1s orbital with the opposite spin ofthe first electron or it could go into the next orbital in the n= 2 level. The next atom is helium with 2 electrons. The orbital diagram, the electron configuration andthe energy diagram. So we have three ways to represent the electron arrangementin an atom. A portionof the energy level diagram is shown, We can alsodisplay the energy level diagram for the hydrogen atom. Theelectronic configuration for hydrogen can be written as 1s 1.This is a short-hand notation which identifies the level, thesublevel and the number of electrons in the sublevel. This notation uses a box to represent the orbital, the labelfor the orbital and an arrow to represent the electron. The orbital diagram for hydrogencan be represented in the following way. Writing the electronconfiguration requires that we recall how many orbitals arecontained in each type of sublevel For example hydrogen with one electron has an electronconfiguration of 1s 1. To designatethe electron configuration we use the level number and the letterof the sublevel and a superscript number to represent the numberof electrons contained in the sublevel. And we use the Pauli exclusionprinciple and other rules to arrange the electrons. 
Arrangements of electrons in the orbitals of an atom iscalled its electron configuration. However, it's easy to determine the configuration of electrons for heavier elements by making a chart.Arrangements of electrons in the orbitals of an atom is called its electron configuration. If there are more electrons than protons, the ion has a negative charge and is called an anion.Įlements are shown from atomic number 1 (hydrogen) up to 94 (plutonium). If there are more protons than electrons, an atomic ion has a positive charge and is called a cation. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons-or not.Īn ion of an atom is one in which the number of protons and electrons is not the same. Remember, a neutral atom contains the same number of protons and electrons. 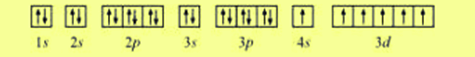
The upper right side shows the number of electrons in a neutral atom. The element atomic number and name are listed in the upper left. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The electron shells are shown, moving outward from the nucleus. Here are electron shell atom diagrams for the elements, ordered by increasing atomic number.įor each electron shell atom diagram, the element symbol is listed in the nucleus. For that, we have electron shell diagrams. It's easier to understand electron configuration and valence if you can actually see the electrons surrounding atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
